Share and Follow
Jade Hlucshniow was on the verge of selling her house to fund treatment for her daughter, whose severe skin condition had robbed her of a normal childhood.
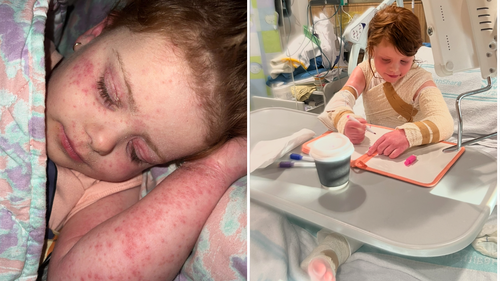
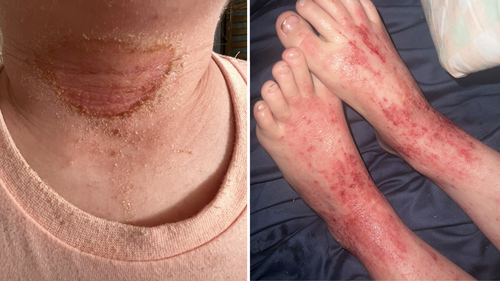
Ruby, now seven years old, endured such intense eczema that it became the focal point of their family’s existence.
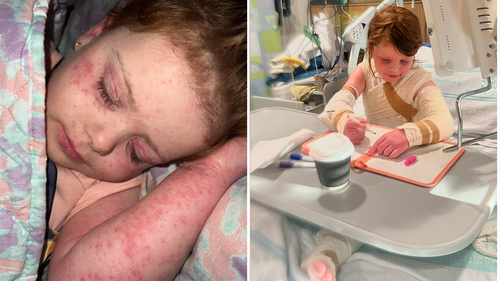
“The nights were the hardest. I will never forget her cries—pleading for relief—and the profound helplessness we felt, unable to ease her suffering,” Jade recounted.
Before Ruby received adequate treatment, Jade described their daily life as a relentless “nightmare.”
Ruby missed school, lost her appetite, and became too self-conscious to venture outside.
“She was so worried about people staring at her,” Jade said.
When the family heard about Dupixent, a treatment Jade describes as a “miracle” injection, the hope came with a brutal price tag: about $1600 a month.
“We found ourselves going over everything – what we could cut from our expenses, how we could earn more money, what we could sell – just to try and make this treatment an option,” explained.
“I can honestly say we would have gone as far as selling our house.”
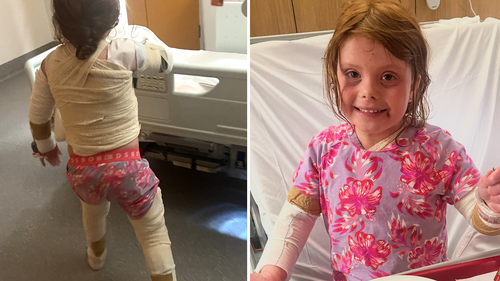
Ruby gained compassionate access to the drug a year and a half ago, and according to her mum, has become “a different child.”
“I can’t put into words the overwhelming relief we felt in that moment…for the first time in so long, we could finally breathe again.”
Now, families who cannot afford the same treatment want the Federal Government to take immediate action by subsidising Dupixent through the PBS for children under 12.
The drug is already available on the Pharmaceutical Benefits Scheme for Australians aged 12 and over, but younger children are excluded without special access.

Managing director Melanie Funk is urging for improved access to treatments, saying the current lack of funding leaves thousands of families suffering unnecessarily.
“Families of young Australian children with severe eczema are at a breaking point, unable to afford a medicine that can change lives,” she said.
If listed on the PBS, the cost would be slashed from $1600 to about $25 a month.
Eczema affects approximately three million Australians, including one-in-three children aged six years or younger.
Dr Li-Chuen Wong, from the Australasian College of Dermatologists, said severe cases could be debilitating for young children, causing relentless itching, skin infections and sleep disruption.

“In the worst cases, children are in constant discomfort and unable to function normally, and the impact on families is profound,” she said.
That was the harrowing reality for the Hlucshniow family – a stark contrast to Ruby’s life today, where she can finally enjoy the same experiences as other seven-year-olds.
“She joins in sports, participates in all school activities, and no longer has to sit on the sidelines. She goes to birthday parties – something she used to avoid – and has become such a social butterfly. It’s been incredible to watch.”
“These might seem like small things, but for Ruby, they mean everything,” Jade said.
The Pharmaceutical Benefits Advisory Committee will consider an updated proposal at its July 2026 meeting.
NEVER MISS A STORY: Get your breaking news and exclusive stories first by following us across all platforms.